With the aim of understanding and recapitulating cellular interactions of hepatocytes in their physiological microenvironment and to generate an artificial 3D in vitro model, a co-culture system using 3D extrusion bioprinting was developed. A bioink based on alginate and methylcellulose (algMC) was first shown to be suitable for bioprinting of hepatocytes; the addition of Matrigel to algMC enhanced proliferation and morphology of them in monophasic scaffolds. Towards a more complex system that allows studying cellular interactions, we applied core–shell bioprinting to establish tailored 3D co-culture models for hepatocytes.
Research Objectives
The aim of the present study was to establish a 3D core–shell bioprinting-based concept for the biofabrication of liver models using algMC as the base bioink to provide stable core and shell compartments supporting encapsulated cells in each phase. Using HepG2, a carcinoma-derived immortalized liver cell line as a model system, we developed an optimized ink in order to better recapitulate their respective biochemical microenvironment and, therefore, support cellular function. This was achieved by functionalizing our previously developed algMC blend with Matrigel. The applicability of the developed ink and core–shell printing in combination with HepG2 cells towards 3D liver models was evaluated. As a further step in the direction of tissue complexity, a fibroblasts-HepG2 co-culture system was established by using coaxial extrusion. After cultivating the printed core–shell constructs for several weeks, cell–cell interactions were studied through evaluation of cellular morphology and distribution as well as expression of relevant biomarker proteins to evaluate the suitability of the developed 3D model to study the influence of the microenvironment on the phenotype and performance of hepatocytes.
Printers and consumables for research institutes
In this work, the extrusion-based technique of 3D plotting was used. Scaffolds were fabricated using pneumatic BioScaffolder 3.1 from GeSiM (Radeberg, Germany) under sterile conditions. The pasty bioinks were dispensed through conical dosing needles (Nordson EFD, Germany) using compressed air; strands were deposited in a layer-by-layer fashion, with parallel alignment in each layer and a shift in orientation of 90° between the layers, in 12 well-plates using air as plotting medium.
Research Process
Viability of HepG2 in algMC printed constructs with and without Matrigel
In order to investigate the viability of encapsulated HepG2, bioprinted and crosslinked constructs were cultured for at least 10 days. At different time points of culture, live and dead cells were simultaneously stained in the scaffolds with Calcein AM and EthD-1.
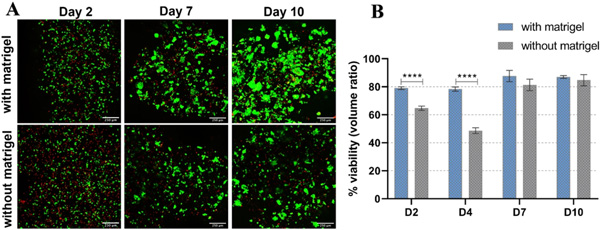
Viability of HepG2 embedded in algMC with and without Matrigel. (A) cLSM images of live/dead staining of HepG2 embedded in printed scaffolds at day 2, 7, and 10 of cultivation. Viable cells stained green (calcein), whereas dead cells are stained red (ethidium homodimer-1); scale bar = 250 µm. (B) Percent viability (volume ratio of viable vs. total clusters) comparing the two conditions (n = 6, mean ± SD, ****p < 0.0001).
Morphology and organization of HepG2 in algMC printed constructs with and without Matrigel
For visualization of the morphology and organization of the cells within the bioprinted hydrogel scaffolds, actin cytoskeleton and nuclei were stained at different time points of culture.
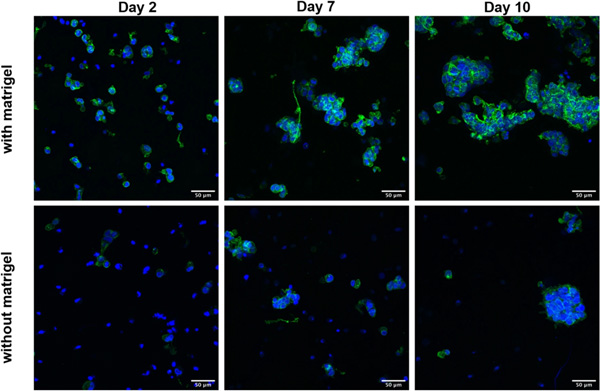
Morphology and organization of HepG2 embedded in algMC with (upper panel) and without Matrigel (lower panel) after 2, 7 and 10 days of cultivation. Cell nuclei are stained in blue (DAPI) while cytoskeletons appear in green (actin staining with Phalloidin-iFluor 488); scale bars represent 50 µm.
Spatially defined pattern of indirect hepatocyte-fibroblast co-culture in a core–shell bioprinted system
After determining a suitable hydrogel material that supports hepatocytes growth and function in a 3D environment, we aimed to investigate their performance in complex co-culture systems fabricated by core–shell 3D bioplotting.
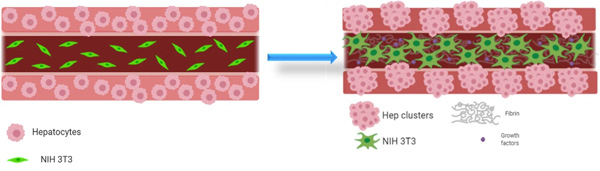
Graphical representation of the hepatocyte-fibroblast co-culture concept, showing hepatocytes encapsulated in the shell are coaxially printed with NIH 3T3 fibroblasts in the core, both co-printed as single cells in their respective bioink (left). An influence of the core composition (with or without fibroblasts and matrix components) on hepatocyte phenotype is hypothesized as they might grow into clusters within their shell compartment and fibroblasts spread to form networks over the cultivation period (right). Images created using Biorender.com software.
Fabrication of cell-laden core–shell strand scaffolds
Core–shell strands were fabricated using coaxial needles of 800 µm (shell) and 400 µm (core) outlet diameter.
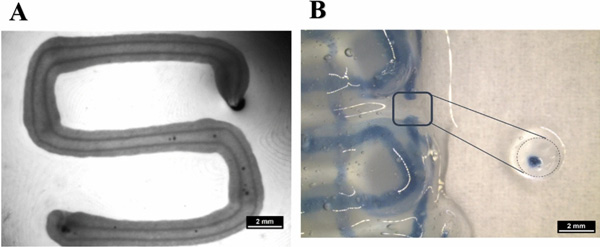
Core–shell plotting using 800 µm needle for shell and 400 µm needle for core. (A) Stereo microscopic image of a cell-free core–shell strand, deposited in meandering shape, with clear separation of core (algMC) and shell (algMC + Matrigel) compartments within the strand; scale bar 2 mm. (B) Stereo microscopic image of a cell-free, printed scaffold with blue stain in the core for visualizing the continuous core compartment through the coaxial strand; The insert shows a section through the strand segment, cut out from the scaffold scale bar 2 mm.
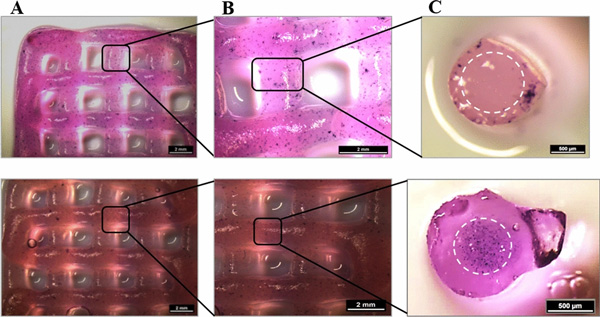
Metabolic activity at day 7 of cultivation (via MTT stain; violet) of HepG2 and NIH 3T3 cells embedded in core–shell printed hydrogel scaffolds; the core–shell interface is visualized via white dashed lines in C. Upper tile shows HepG2 encapsulated in the shell compartment consisting of algMC + Matrigel whereas the core remained cell free. Lower tile shows NIH 3T3 cells encapsulated in the core compartment consisting of algMC whereas the shell remained cell-free. (A,B) Scale bars 2 mm. (C) Scale bars 500 µm.
Simultaneous embedding of HepG2 and NIH 3T3 cells in core–shell strand scaffolds
In order to visualize both cell types after simultaneous bioprinting, first co-culture experiments were conducted with pre-labeled cells: DiD-labeled hepatocytes (red) were encapsulated in shell compartment consisting of algMC + Matrigel while DiI-labeled fibroblasts (cyan) were encapsulated in the core consisting of algMC. These scaffolds were then compared to monoculture scaffolds which had DiD-labeled hepatocytes in shell only with no cells in the core.
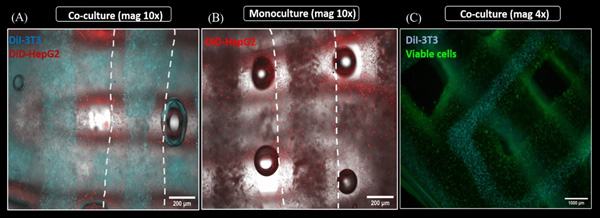
Spatially defined distribution of HepG2 and NIH 3T3 in core–shell strand scaffolds. (A,B) Fluorescence images of strands in a scaffold illustrating distribution of encapsulated cells in HepG2/NIH 3T3 co-culture vs. HepG2 monoculture scaffolds; scale bar 200 µm. White dashed lines indicate the whole strand width. (C) Fluorescence image of core–shell strand co-culture scaffolds showing Calcein-stained viable cells in green on day 4 of cultivation; for visualization of the core, the cyan channel (DiI-labeled NIH 3T3) was overlaid; scale bar 1000 µm.
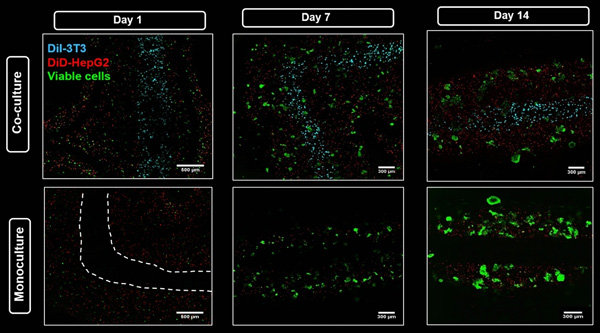
Viability and localization of HepG2 in co-culture with NIH 3T3 and in monoculture in core–shell strand scaffolds. Confocal images show DiD-labeled HepG2 (red) encapsulated in shell compartment consisting of algMC + Matrigel with either NIH 3T3 fibroblasts encapsulated in algMC core and labeled with DiI (cyan) (upper tile) or with a cell-free algMC core (lower tile). Viable cells were stained with Calcein-AM (green). Figure shows the progression of cell cluster formation of hepatocytes over the culture period. Scale bar (day 1) 500 µm; scale bar (days 7 and 14) 300 µm; white dashed line indicates here the core–shell interface.
Functionalization of core bioink – enhancing fibroblast network formation
As we hypothesized that the phenotype of the fibroblasts has a direct impact on the interaction with the hepatocytes, the next step was investigating the effect of functionalized bioinks on NIH 3T3 phenotype.
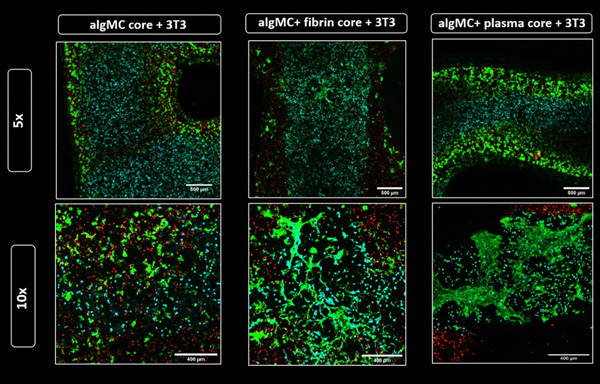
Fibroblasts network formation in the core compartment at day 7 of co-culture. Confocal images of core–shell hydrogel strands with encapsulated DiD-labeled HepG2 (red) in algMC + Matrigel shell and DiI-labeled NIH 3T3 fibroblasts (cyan) embedded in the core of algMC, algMC supplemented with fibrin and algMC supplemented with plasma. Viable cells are stained green with Calcein AM, indicating formation of fibroblast networks within the fibrin- and plasma-functionalized core while a round morphology of the fibroblasts is maintained in unmodified algMC core. Upper tile gives an overview of a section of the core–shell strand; magnification 5x, scale bar 500 µm. Lower tile shows higher magnification (10x) images of NIH 3T3 fibroblasts in the core; Scale bars 400 µm.
Influence of the microenvironment on expression of hepatic marker proteins in the core–shell bioprinted co-culture system
Hepatocytes functionality is often assessed by their ability to synthesize and secrete specific proteins. Therefore, in order to investigate the effect of the fibroblasts grown in different core materials on the hepatocytes in the neighboring shell compartment, the expression of specific marker proteins in the different conditions was observed by antibody staining and subsequent immunofluorescence microscopy.
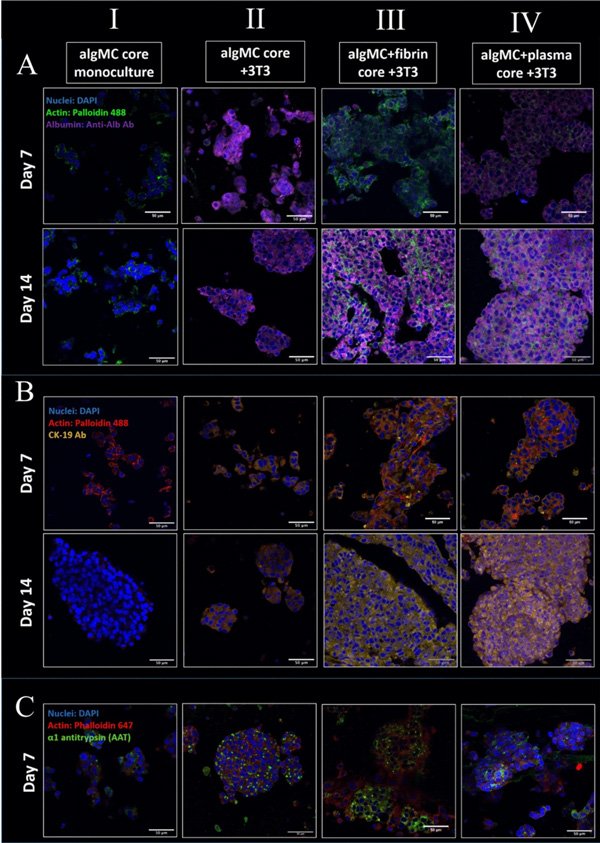
Cluster formation and biomarker expression of HepG2 embedded in shell compartment in monoculture (cell-free algMC core; I) and in co-culture with NIH 3T3 fibroblasts embedded in core compartment of different compositions (II-IV). (A) Confocal images of HepG2 stained for Albumin (purple), nuclei (blue) and cytoskeletons (green); scale bars 50 µm. (B) Confocal images of HepG2 stained for CK-19 (yellow), nuclei (blue) and cytoskeletons (red); scale bars 50 µm. (C) Confocal images of HepG2 stained for AAT (green), nuclei (blue) and cytoskeletons (red); scale bars 50 µm.
核壳生物打印系统中肝细胞-成纤维细胞间接共培养的空间定义模式
In order to further characterize the modified hydrogel blends regarding their post-printing and -crosslinking properties, uniaxial compressive tests were performed for monophasic scaffolds to study the mechanical stability and stiffness of 3D constructs after plotting and post-processing.
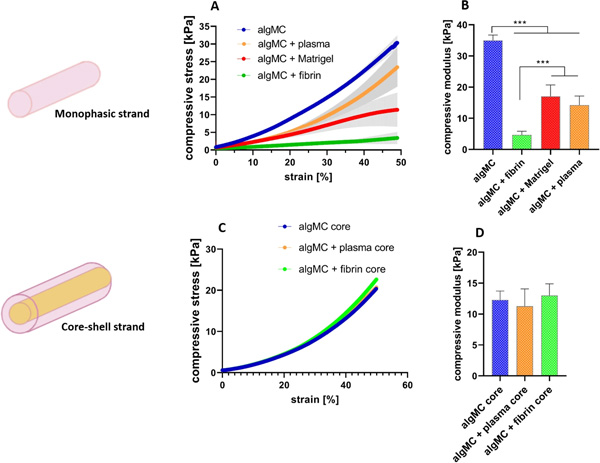
Mechanical characterization of monophasic and core–shell strand scaffolds after printing and crosslinking. (A) Stress–strain curves of cross-linked monophasic scaffolds at day1. (B) Compressive modulus of cross-linked monophasic scaffolds at day 1. (C) Stress–strain curves of cross-linked core–shell scaffolds with varying core compositions in algMC + Matrigel shell at day1. (D) Compressive modulus of cross-linked core–shell scaffolds at day 1 (n = 4, p*** < 0.0005, mean ± SD).
With the aim of fabricating liver tissue models, co-culturing fibroblasts in core–shell fashion was the first step to provide insights into the great potential of this co-culture bioprinting strategy. Towards developing in vitro liver models, the development of co-culture systems with other cell types would be the aim of our future work after demonstrating here how using core–shell technology enabled us to engineer a tailored 3D microenvironment for the bioprinted cells. In order to mimic the physiological liver structures which constitutes of vasculature and interconnected vessels, the next steps would be to further increase the complexity developing printed constructs for co-cultivation of hepatocytes with endothelial cells and other supporting cell types. This will allow us to study the cellular interactions in these 3D systems and to establish perfusable liver models.
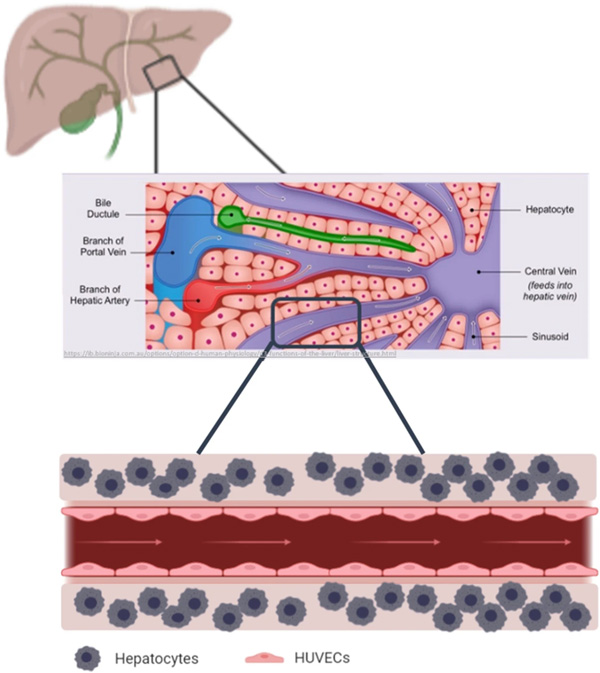
Graphical representation of the hepatocyte co-culture concept, showing how an in vivo liver lobule is consisting of blood vessels, ducts and canals (vasculature) that are surrounded by sheets of hepatic cells as shown in the upper illustration. Therefore, the concept of mimicking the microarchitecture of the liver lobule is envisaged to be represented in a coculture model of hepatocytes printed in the shell and endothelial cells lining the core of a perfusable construct (lower illustration). Middle illustration obtained from Cornell, B. 2016. Hepatic lobules.
Research Conclusion
In this work, we have presented 3D extrusion-based bioprinting as a promising tool to generate biologically active liver tissue models. By specific adaptation of the bioink composition and by applying coaxial printing of two distinct bioinks in core–shell fashion, the 3D microenvironment of hepatocytes can be controlled and tuned to mimic specific conditions. With the presented method, we established a functional co-culture model of hepatocytes and fibroblasts with a core–shell strand/scaffold design. By comparing different ink compositions (algMC with and without bioactive components) and co-cultures vs. monocultures, we could demonstrate in our proof-of-concept study a strong influence of changing components of the spatially defined 3D microenvironment on hepatocyte proliferation and expression levels of selected hepatocyte-specific markers. These findings highlight the importance of tailoring the 3D microenvironment towards generation of artificial liver constructs and the high potential of versatile bioprinting technologies for that purpose.























 Home
Home Telephone
Telephone Message
Message







